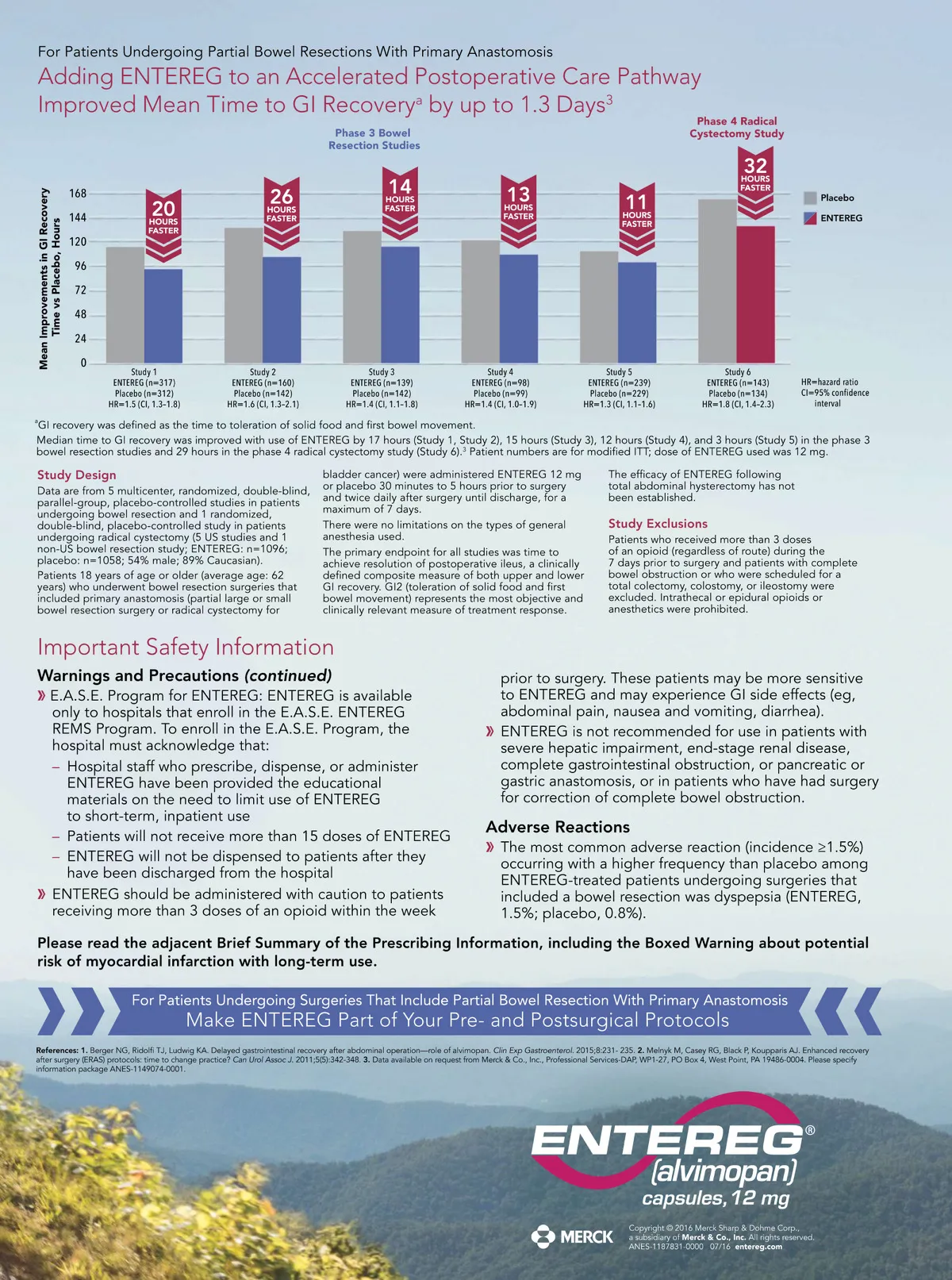
In clinical trials, ENTEREG added to an accelerated care pathway (ACP), also commonly called an enhanced recovery pathway (ERP), 1,2 was more effective than an ACP alone in helping to ACCELERATE GI RECOVERY The ACP used in clinical trials included: Early diet advancement Early nasogastric tube (NGT) removal Early ambulation Indication and Usage ENTEREG is indicated to accelerate the time to upper and lower gastrointestinal recovery following surgeries that include partial bowel resection with primary anastomosis. Important Safety Information WARNING: POTENTIAL RISK OF MYOCARDIAL INFARCTION WITH LONG-TERM USE: FOR SHORT-TERM HOSPITAL USE ONLY Increased incidence of myocardial infarction was seen in a clinical trial of patients taking alvimopan for long-term use. No increased risk was observed in short-term trials. Because of the potential risk of myocardial infarction, ENTEREG is available only through a restricted program for short-term use (15 doses) called the ENTEREG Access Support and Education (E.A.S.E.) Program. Contraindications ENTEREG Capsules are contraindicated in patients who have taken therapeutic doses of opioids for more than 7 consecutive days immediately prior to taking ENTEREG. Warnings and Precautions There were more reports of myocardial infarctions in patients treated with alvimopan 0.5 mg twice daily compared with placebo-treated patients in a 12-month study of patients treated with opioids for chronic pain. In this study, the majority of myocardial infarctions occurred between 1 and 4 months after initiation of treatment. This imbalance has not been observed in other studies of alvimopan, including studies of patients undergoing bowel resection surgery who received alvimopan 12 mg twice daily for up to 7 days. A causal relationship with alvimopan has not been established.
Journal of the American College of Surgeons Volume 226 Issue 6 June 2018: Page 2